With the wide spread outbreak of life-threatening Covid-19 (Corona Virus) all across the world, there is growing demand for fast and reliable detection.
EWC Diagnostics and our Production partners offer you: Swabs, VTM/CDC/PBS medium, Rapid Test (15 minutes), PCR and RNA extraction. Next to that we can offer you automated solutions like the KingFisher, Roboprep and InstaNx. Please find the product details of the different tests on this page.
We have Corona tests on stock, welcome to contact us at any time ( sales@ewcdiagnostics.com )!
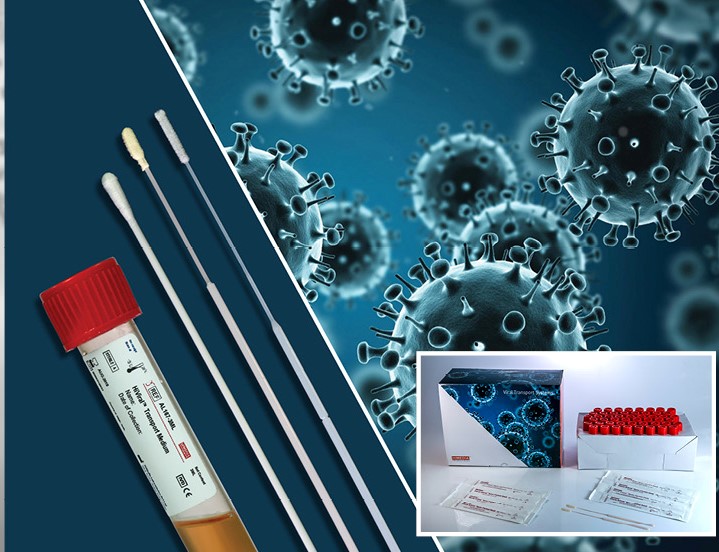
Virus Transport Kit
The Virus Transport Kit is a specially designed transport system to collect and transport viruses in an active form to the laboratory for isolation. It is designed to maintain the viability and the virulence of the viral sample.
Virus Transport Medium (VTM Virus Transport Medium for Viruses, Chlamydia, Mycoplasma and Ureaplasma) is made of Hanks Balanced Salt Solution and contains a protective protein, antibiotics to control microbial and fungal contamination and buffers to control the pH. Phenol red is used as a pH indicator. The medium also contains a cryoprotectant which helps in preserving the viruses if specimens are frozen for prolonged storage.
We offer nasal and throat swabs together with the Transport Medium. The flocked nylon swab (nasopharyngeal swab) has a short perpendicular ultra-flexible plastic shaft that is designed for better patient comfort.
EWC Diagnostics offers different Transport Medium recipes: VTM, CDC (WHO formulation), PBS.
Corona PCR test kit
SARS-CoV-2 is a test based on real-time reverse-transcription and amplification designed for qualitative determination of absence or presence of SARSCoV-2 in a RNA extract. This test is indicated to diagnose the occurrence of this infection in humans, or complement a proven or indeterminate diagnosis.
• PCR Targets R-IP2Gene, R-IP4 Gene, N Gene.
• RT PCR design, WHO registered
• Validated on the following specimen: Nasopharyngeal aspirate, Bronchoalveolar fluid, Sputum and Nasal swab.
• Encapsidated control of RNA extraction and RT-PCR inhibition
• Validated on CFX96TM, LightCycler®480 Instrument II (Roche), Applied Biosystems® 7500 Real-Time PCR Systems, QuantStudio 5® (Applied Biosystems).
Corona RNA extraction kit
Patient RNA samples can be extracted and purified using the Magnetic Preparation Kit from upper and lower respiratory specimen swabs (such as nasopharyngeal or oropharyngeal, sputum and bronchoalveolar lavage fluid) obtained from individuals suspected of having respiratory tract infections.
Principle:
• Lysis step using enzymes, carrier tRNA and lysis buffer.
• Extracted RNA or DNA is then bound to the magnetic beads and purified with the wash buffers to remove protein and other cellular contaminants.
• Purified RNA or DNA is eluted in an appropriate elution buffer and is then ready to be used in PCR detection kits.
Features:
• Innovative magnetic bead technology for the isolation of highly purified RNA/DNA.
• Optimized and validated for a wide range of viruses (e.g. Coronaviridae, Orthomyxoviridae, Asfarviridae, Picornaviridae, and many more).
• High throughput with automation solutions.

